Aesthetic clinics handle two of the most heavily regulated data types in cosmetic medicine — patient consent and before/after photographs. Done right, they're trust signals and marketing assets. Done wrong, they're an audit risk and a liability lawsuit waiting to happen.
1Why paper consent forms are a liability in 2026
Most Australian aesthetic clinics still rely on paper folders for treatment consents. It feels familiar, but in 2026 it's a real risk — and any AHPRA-registered practitioner reviewing your records knows it.
- Lost or shredded too soon. Legal retention for cosmetic-procedure records is typically 7+ years (longer for minors). A misfiled folder is a compliance gap.
- No timestamp, no version control. When a complaint is raised two years later, proving exactly which version of the consent the patient signed becomes nearly impossible.
- Hard to retrieve when an incident occurs. An auditor or insurer wants the record in minutes, not after a basement search.
2What digital consent should capture
A compliant digital consent record — the kind your insurer and AHPRA expect for cosmetic procedures — should capture every one of the following. TGA-aware language matters too: never describe a Schedule 4 cosmetic injectable by brand name in patient-facing materials.
- Patient's full name — typed signature is acceptable in most Australian jurisdictions; a drawn signature is best practice for higher-risk treatments.
- Treatment name and the version of the consent form the patient agreed to.
- Immutable timestamp — once written, it cannot be edited.
- Practitioner name — who took the consent and whether they were AHPRA-registered at the time.
- Audit trail — who viewed or exported the record, and when.
3Before/after photos — your best marketing tool, your biggest legal risk
Before/after images are the single most powerful marketing asset an aesthetic clinic owns. They're also the data type most likely to land you in front of a privacy regulator.
- Photos are special-category health data under the Privacy Act and similar regimes.
- They require explicit, separable marketing consent — clinical use is not marketing use, and bundling them is a documented compliance mistake.
- Storage requirements: encrypted at rest, access controlled, retention period documented.
- You must be able to delete on patient request — the right to erasure applies.
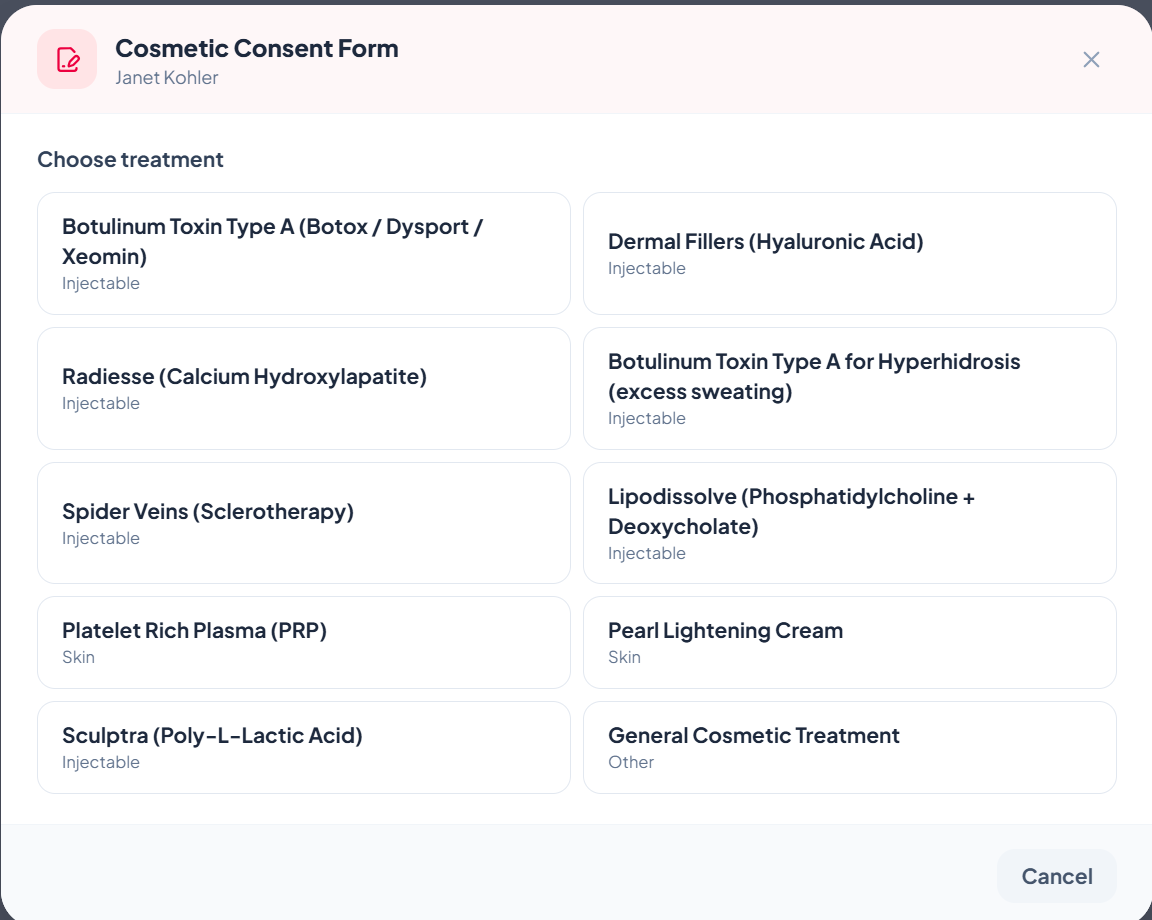
4Inside ZibaDesk's e-signature consent form
ZibaDesk's consent system was modeled directly after the printed consent forms that established Australian aesthetic clinics already use — same structure, same protections, just digitized.
10 ready-to-use treatment templates
We ship pre-built templates so you don't write your own from scratch:
- Botulinum Toxin Type A (Botox / Dysport / Xeomin)
- Dermal Fillers (Hyaluronic Acid)
- Radiesse (Calcium Hydroxylapatite)
- Hyperhidrosis (excess sweating treatments)
- Spider Veins (Sclerotherapy)
- Lipodissolve (Phosphatidylcholine + Deoxycholate)
- Platelet Rich Plasma (PRP)
- Pearl Lightening Cream
- Sculptra (Poly-L-Lactic Acid)
- Generic Cosmetic Treatment (catch-all)
Each template includes 12 general acknowledgements (allergic reactions, infection risk, pregnancy contraindication, post-care responsibilities, cost transparency) plus 4–10 treatment-specific bullets — for example, "I understand Sculptra's effects appear gradually over 6 weeks" for Sculptra.
Three signature pads — patient, practitioner, witness
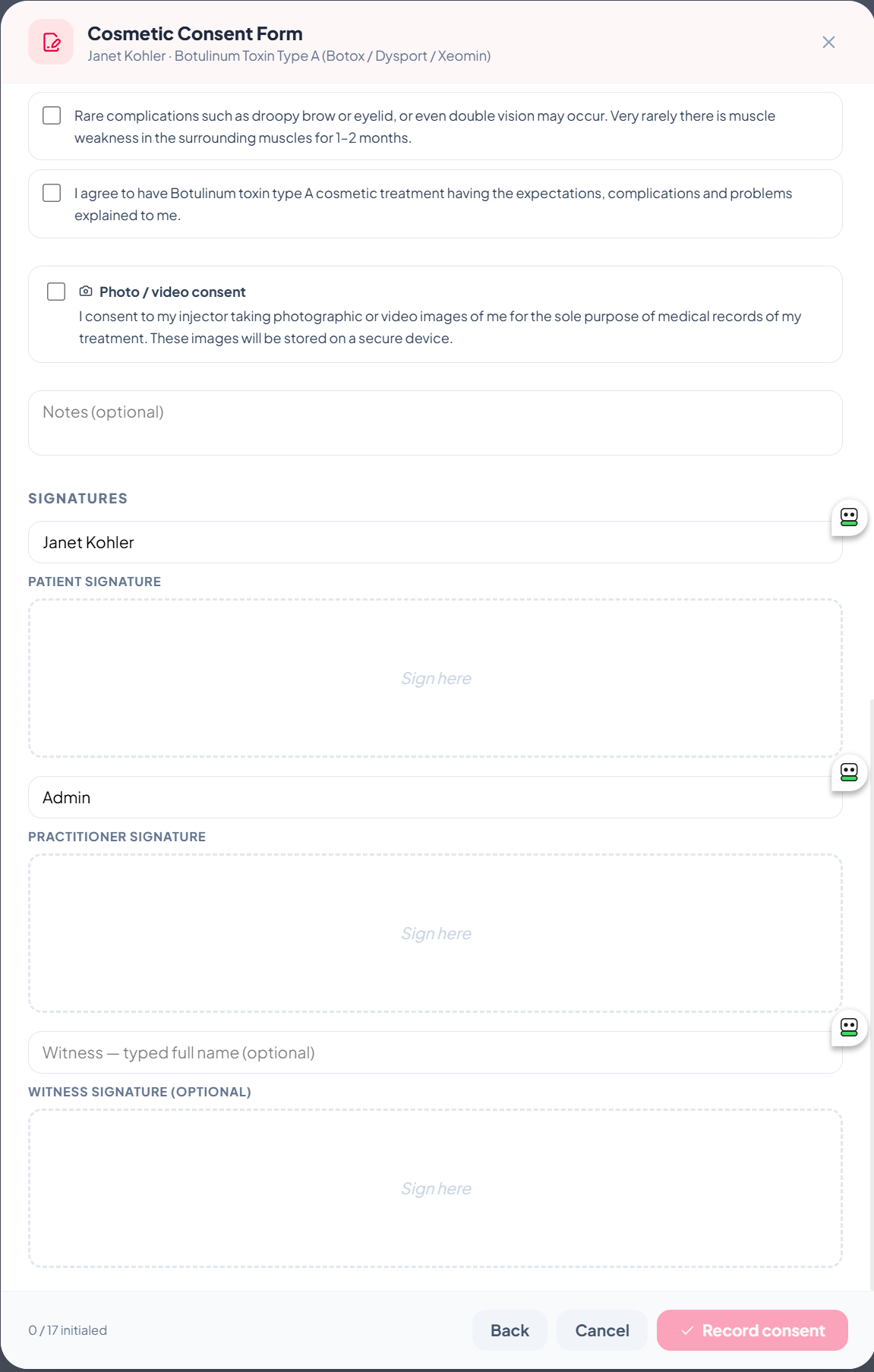
Every consent captures up to three drawn e-signatures, signed directly on the screen with a finger or stylus on iPad, or with a mouse on desktop:
- Patient signature — required, plus a typed full name as audit fallback.
- Practitioner signature — captures the registered nurse or doctor performing the treatment.
- Witness signature — optional, used in higher-risk treatments or when local rules require.
All signatures are stored as PNG images alongside the consent record, with an immutable timestamp.
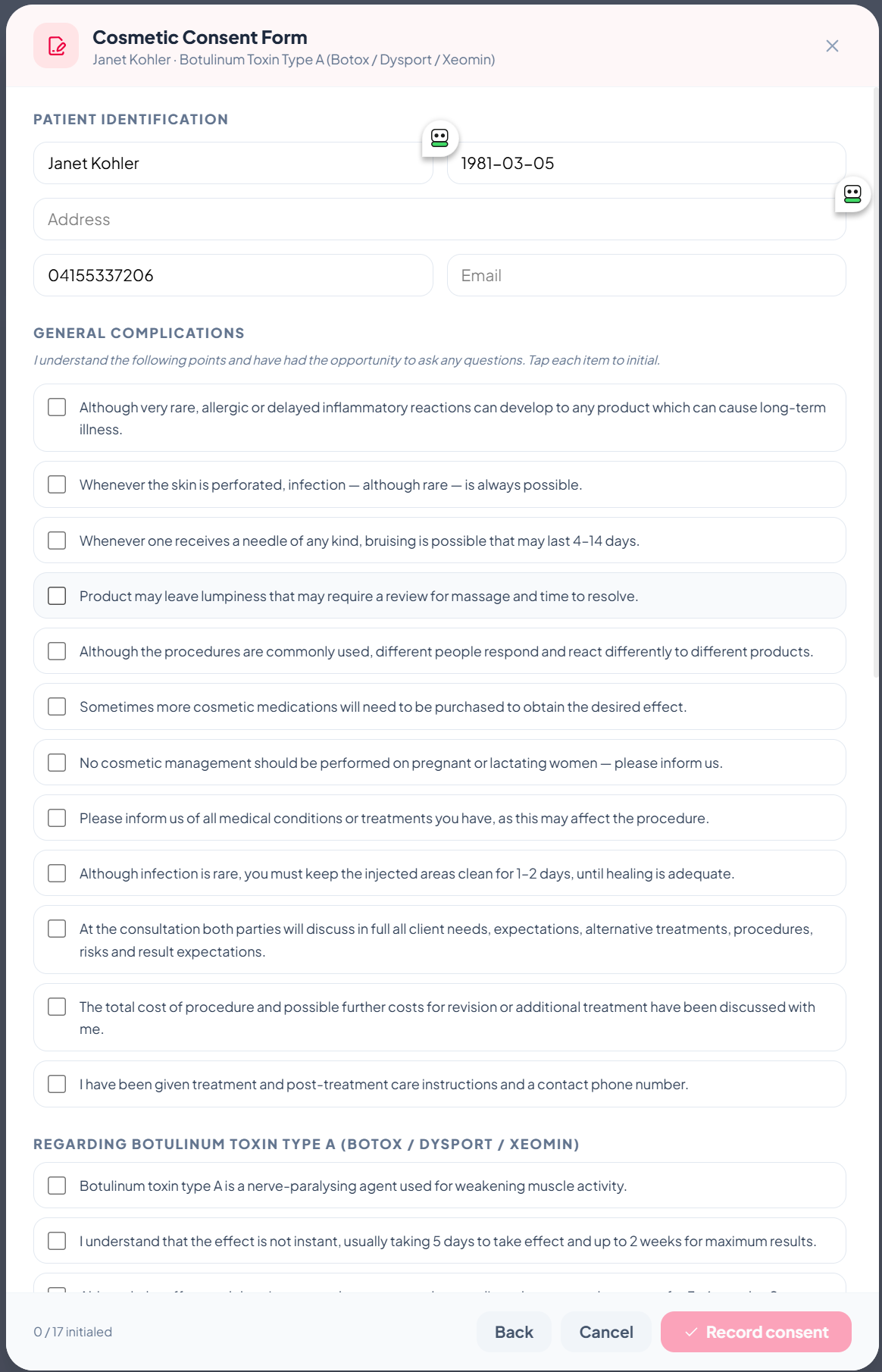
How it works in practice
- Open the customer record → tap "Record new consent".
- Pick the treatment template.
- Patient details (name, DOB, address, phone, email) auto-populate from their existing record — no re-typing.
- Patient taps each acknowledgement to initial.
- Patient signs on the canvas → practitioner signs → optional witness signs.
- Photo/video consent for clinical use is captured as a separate toggle.
- Tap "Record consent" — that's it. The full signed form lives forever on the patient record.
Compliance baked in
- Australia: AHPRA-aware structure (separable clinical vs marketing consent), TGA-conscious S4 medication wording.
- Germany: DSGVO Art. 9 compliant — special-category health data handled with explicit consent + audit trail.
- France: RGPD Article 9 compliant.
- All others: structure follows the WHO informed-consent guidance.
5How ZibaDesk handles photos and storage
Beyond the consent capture itself, the photo and storage layer is built for the same audit-ready standard.
- Before/after photo library with treatment-area tags, before/after/progress labels, a separate marketing-consent toggle on every photo, and a built-in lightbox view for in-treatment-room reviews.
- In-browser compression to roughly 150 KB JPEGs — no patient image ever leaves the device unencrypted.
- Encrypted storage aligned with Australian Privacy Principles, DSGVO, and RGPD requirements for international tenants.
- Per-patient export of the entire consent and photo history in seconds — for insurer requests, auditor reviews, or patient subject-access requests.
6Practical checklist for your clinic
- Move existing paper consents to digital within 90 days.
- Train all staff on the difference between clinical consent and marketing consent.
- Document your retention policy (7+ years for medical records in most jurisdictions).
- Set up automatic, encrypted backups.
- Have a documented patient-data-deletion procedure.